Sabinsa Corporation has been granted permission by the Drugs Controller General of India to manufacture and market Ocufors™, a patented ophthalmic solution containing one percent forskolin, for the treatment of glaucoma. This is the first time that the government of India has approved a product developed from natural sources for use as a pharmaceutical grade drug.
“Research and development has always been strength and focus for Sabinsa, and through our ongoing efforts we were able to develop this stable, water-soluble forskolin solution that has been shown in clinical trials to be 30 percent more effective than the most popular glaucoma treatment on the world market today,” noted Dr. Muhammed Majeed, founder and CEO, Sabinsa Corporation. “It is an honor to receive this approval, which speaks highly about our stringent safety, efficacy and quality standards, which we apply to all of our ingredients whether they be prescription grade, generic commodities or our branded offerings.”
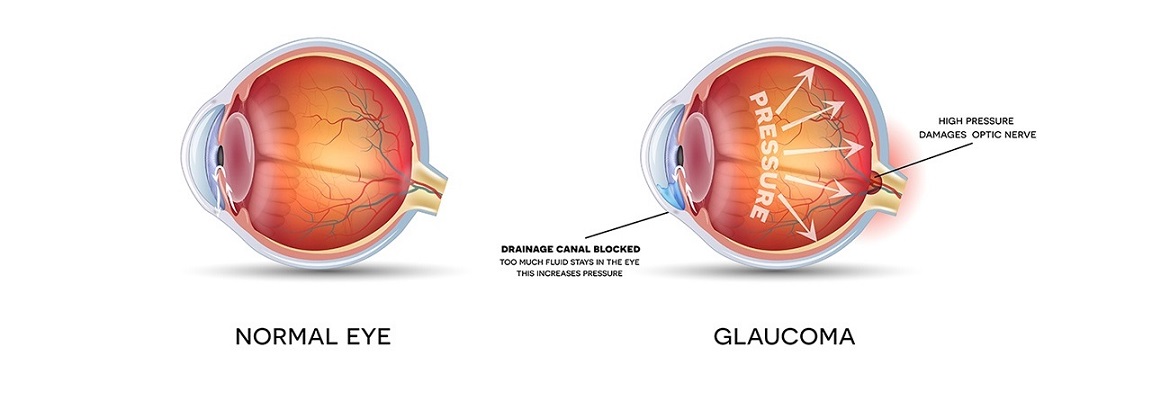
Glaucoma is a degenerative disease of the optic nerve, often caused by increased pressure inside the eye. Glaucoma affects more than 50 million individuals worldwide and is the second leading cause of blindness across the globe. Ocufors, which is administered as an eye drop, offers an effective treatment for glaucoma with minimal side effects. Sabinsa is currently seeking registration for Ocufors in the U.S., Europe and Australia.
The active ingredient in Ocufors, forskolin, is derived from the herb Coleus forskohlii, a plant native to subtropical and warm temperate habitats, and a member of the mint family. Sabinsa holds U.S. Patent #6,960,300 and pending international patent applications for this composition.